
Sp and sp2 hybridization results in two and one unhybridized p orbitals respectively whereas in sp3 hybridization there are no unhybridized p orbitals. We're sorry, but in order to log in and use all the features of this website, you will need to enable JavaScript in your browser. * Each of these sp3 hybrid orbitals f… Generally, the Lewis structure is helpful to understand the molecular geometry of any given chemical compound. This intermixing is based on quantum mechanics. These hybridizations are only possible for atoms that have d orbitals in their valence subshells (that is, not those in the first or second period).  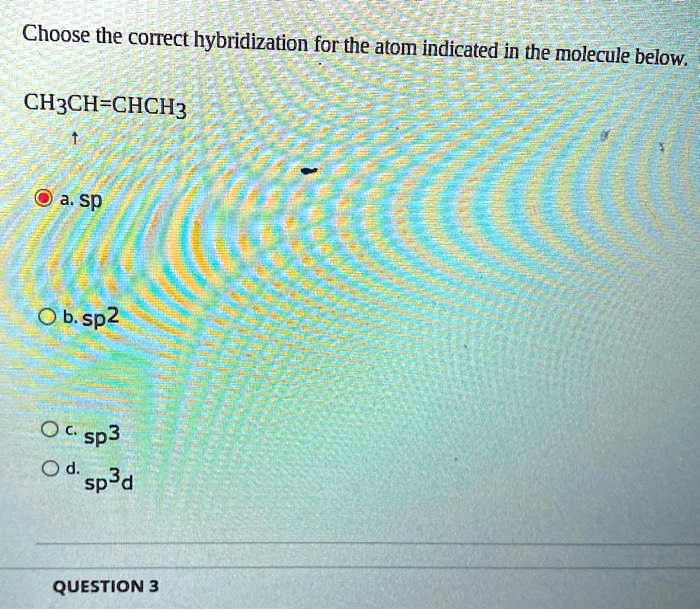
As there are molecules of Iodine, one molecule of Iodinewill be in the centre. Sp 3 d Hybridization sp 3 d hybridization is shown in phosphorus penta chloride (PCl 5 ).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
